First Name:
Derek Last Name:
WangMentor:
Dr. Xuexia WangAbstract:
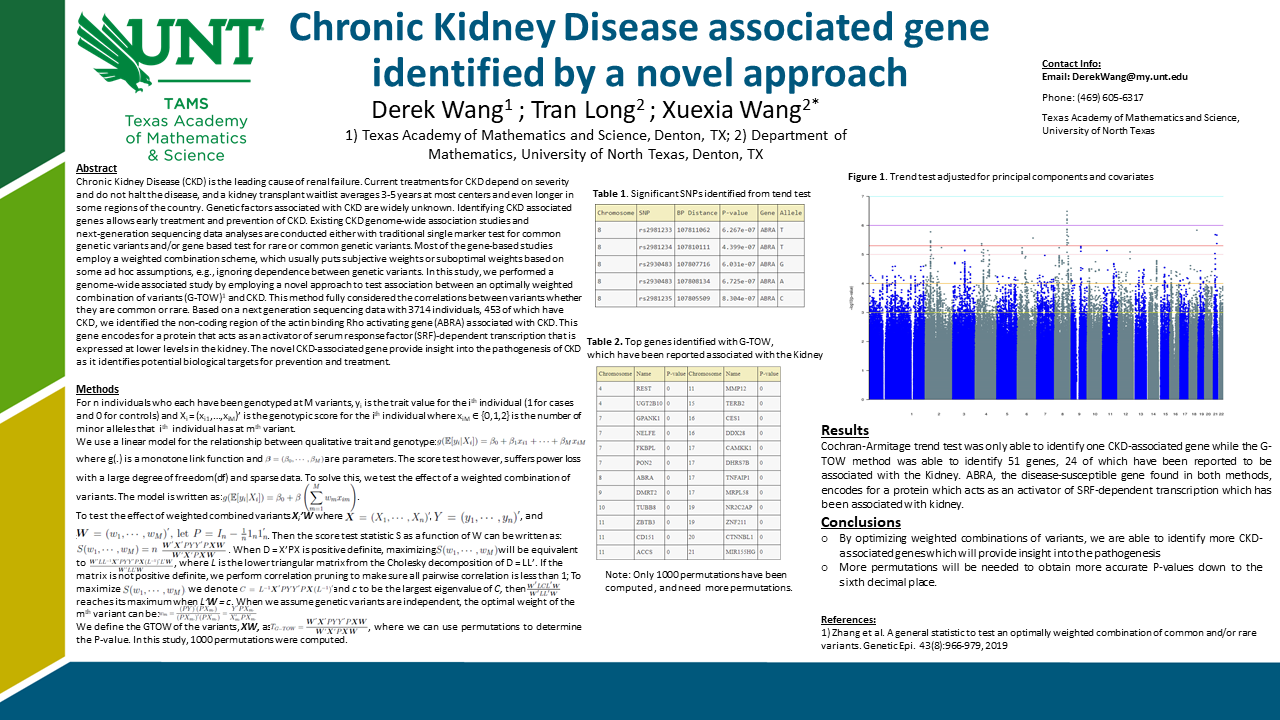
Chronic Kidney Disease (CKD) is the leading cause of renal failure. Current treatments for CKD depend on the severity
and do not halt the disease, and a kidney transplant waitlist averages 3-5 years at most centers and even longer in
some regions of the country. Genetic factors associated with CKD are widely unknown. Identifying CKD-associated
genes allow early treatment and prevention of CKD. Existing CKD genome‐wide association studies and
next‐generation sequencing data analyses are conducted either with traditional single-marker tests for common
genetic variants and/or gene-based tests for rare or common genetic variants. Most of the gene-based studies
employ a weighted combination scheme, which usually puts subjective weights or suboptimal weights based on
some ad hoc assumptions, e.g., ignoring dependence between genetic variants. In this study, we performed a
genome-wide associated study by employing a novel approach to test the association between an optimally weighted
combination of variants ( G‐TOW) 1 and CKD. This method fully considered the correlations between variants whether
they are common or rare. Based on a next-generation sequencing data with 3714 individuals, 453 of which have
CKD, we identified the noncoding region of the actin-binding Rho activating gene (ABRA) associated with CKD. This
gene encodes for a protein that acts as an activator of serum response factor (SRF) dependent transcription that is
expressed at lower levels in the kidney. The novel CKD-associated gene provides insight into the pathogenesis of CKD as it identifies potential biological targets for prevention and treatment.Poster:
Year:
2021



